India Pharma Sector
India’s pharmaceutical sector—long dubbed the “Pharmacy of the World”—is undergoing a dynamic transformation to strengthen its global standing, ensure domestic self-reliance, and boost public health preparedness. With strategic policy interventions, technological innovations, and industry reforms, this overhaul offers critical insights for UPSC aspirants, covering economy, health, governance, and technology.
Sector at a Glance
- Market size: Valued at approximately US $50 billion in FY 2023–24, projected to reach US $130 billion by 2030.
- Generics dominance: Supplies 20% of global generic demand by volume; more than 50% of global vaccines.
- Export leadership: Exports reached around US $27.8 billion in FY 2023–24, serving regulated markets like the US, UK, EU, and Canada
- Growth trajectory: India posted a 7.8% year-on-year increase in pharma output in April 2025, signaling sectoral vigor
Policy & Reforms: Building Resilience
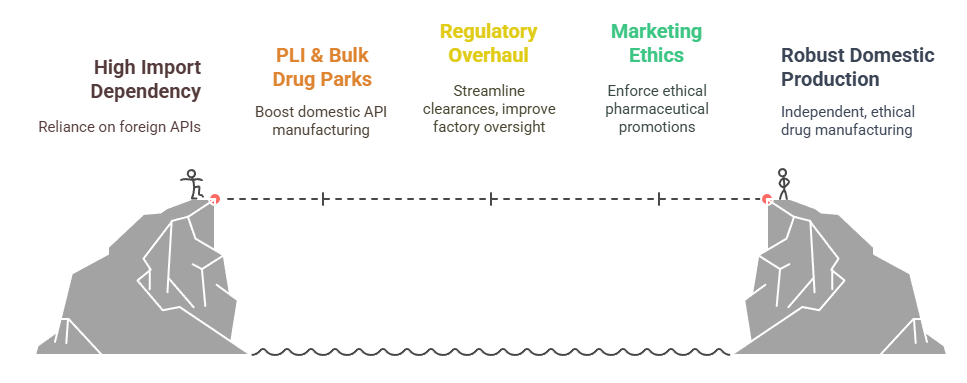
a) PLI & Bulk Drug Parks
Initiatives like the Production Linked Incentive (PLI) schemes and bulk drug parks have been central to boosting domestic API manufacturing and reducing import dependency on China
b) Regulatory Overhaul
The Drug Controller General of India (DCGI) has embarked on reforms—including digital clearances and export simplification—to halve NOC wait-time and improve oversight of 10,000+ pharmaceutical factories
c) Marketing Ethics
In March 2024, the Uniform Code of Pharmaceutical Marketing Practices (UCPMP) was introduced, evolving into a statutory standard by September to clamp down on unethical promotion
Manufacturing Hubs & Investments
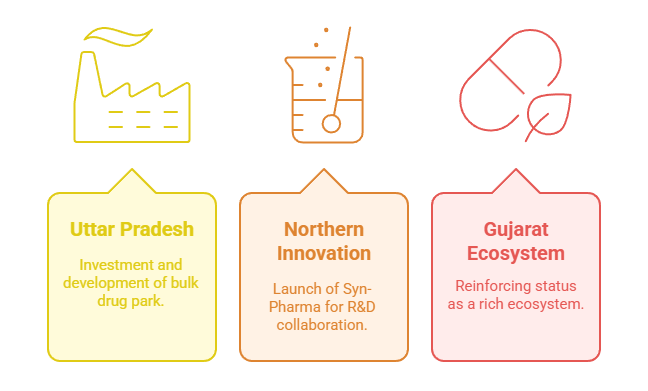
a) Uttar Pradesh’s Pharma Push
UP inked Rs 435 crore MoUs at Ahmedabad’s investment roadshow, including a 1,472-acre bulk drug park in Lalitpur and new formulation plants—reinforcing India’s ambitions of self-reliance
b) Northern Pharma Innovation
PI-RAHI launched Syn-Pharma at PU Chandigarh to foster academia-industry R&D collaboration, offering grants and PhD programs in drug development.
c) Gujarat’s Drug Ecosystem
Gujarat—with over 3,500 drug units and multiple bulk drug parks—continues to reinforce its status as a rich pharmaceutical ecosystem .
Innovation & Technology Adoption
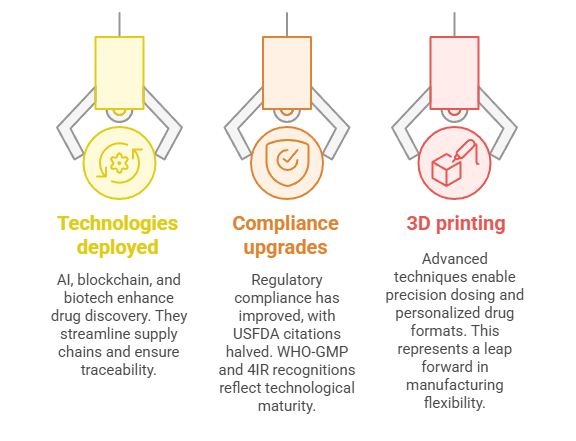
- AI, blockchain, biotech: Technologies are being deployed to enhance drug discovery, streamline supply chains, and ensure traceability—key to export resilience.
- Compliance upgrades: A McKinsey report underscores India’s improved regulatory compliance—USFDA “OAI” citations halved over a decade; WHO-GMP and 4IR lighthouse recognitions reflect technological maturity.
- 3D printing: Advanced techniques are emerging, enabling precision dosing and personalized drug formats—a leap forward in manufacturing flexibility.
Access, Affordability & Public Health
- Jan Aushadhi scheme: Over 9,000 kendras provide generic medicines at 50–90% lower prices, saving citizens an estimated ₹15,000 crore in FY 24–25
- Demand surge & quality scrutiny: Rampant use of generics has reduced health costs, but incidents—like the Gambia-linked cough syrup controversy—highlight the need for stringent quality guards
Export Strategy & Global Outreach
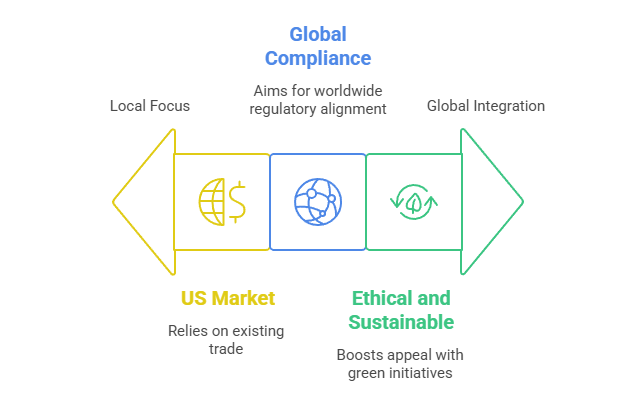
- U.S. market dependence: Explains ~30% of India’s pharma exports (~US $8.7 billion). Industry lobbies are pressing for zero import duties to maintain competitiveness.
- Global compliance governance: Proposed shift to ICH/PIC-S standards is aimed at enhancing export acceptance and joining global regulatory regimes within 10–15 years .
- Ethical and sustainable practices: Emphasis on carbon reduction, waste management, and ethical sourcing lifts India’s green credentials—a valuable export differentiator.
Challenges on the Horizon
- API import dependency: Approximately 70% of APIs are imported, primarily from China—posing risk and reinforcing calls for domestic capacity building
- Quality concerns: The cough syrup crisis exposed weaknesses in fragmented regulatory architecture; there’s urgency for a unified regulator to enforce standards
- R&D shortfalls: India spends only 8.4% of pharma sales on R&D, below the global average of 10–11%. Policy demands include enhanced tax incentives, patent-box benefits, and international collaborations
- Skilled workforce & innovation gap: A shortage in biotechnology expertise and advanced manufacturing calls for stronger human capital development strategies .
Strategic Roadmap Ahead
| Strategy Area |
Key Recommendations |
| Domestic API Scaling |
Expand PLI 2.0, develop green API parks, promote fermentation technology |
| Regulatory Reform |
Unify drug regulation; digital-clearance enhancement; ICH/PIC-S adoption |
| R&D Upgradation |
Increase R&D spending; tax incentives; PPP research hubs; AI-led discovery |
| Export Diversification |
Pursue Africa/ASEAN markets; build brand through ethics and green standards |
| Technology Integration |
Deploy AI, blockchain, 3D-printing; boost 4IR and smart-quality frameworks |
Conclusion
India’s pharmaceutical overhaul is both a public health imperative and an economic opportunity. Through smart policymaking, technological adoption, quality improvement, and global market expansion, India is strategically positioning itself as a leader in affordable, innovative, and reliable medicine. |







The focus on innovation and regional contributions is key to India’s pharma success. I think the synergy between policy, innovation, and local manufacturing will play a major role in helping India scale up its exports and take a leadership role globally.