Pharmaceutical Safety and the State: A Sociological Lens on Schedule M Reforms
(Relevant for Sociology Paper 2: Population Dynamics)
|
“Health is not merely the absence of disease — it is a social right, and its violation reflects the structural violence embedded in our institutions.” In response to recent deaths of children linked to cough syrups contaminated with Diethylene Glycol (DEG), the Union Health Ministry has directed the strict implementation of revised Schedule M norms under the Drugs and Cosmetics Act, 1940. These norms are designed to overhaul drug manufacturing standards across India, aligning them with WHO Good Manufacturing Practices (GMP). But beyond the technocratic vocabulary of “compliance,” “risk management,” and “data integrity,” this issue reveals deeper sociological questions:
Public Health as a Social Institution
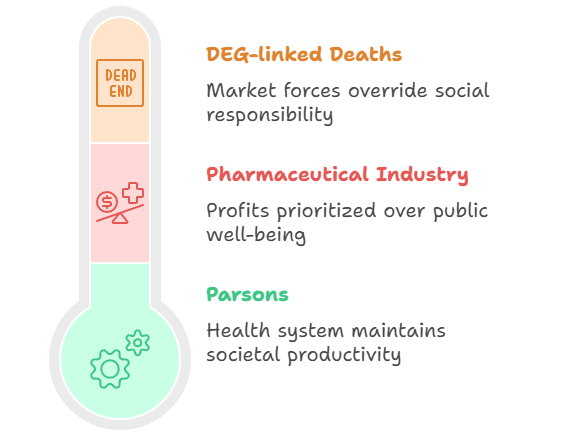
Health is not just a biomedical concern — it is a social product shaped by class, governance, and institutional structures. Talcott Parsons – Sick Role and Health as Functionality: Parsons viewed health systems as functional prerequisites for society — keeping individuals productive. But when the health system itself fails, especially through preventable deaths due to poor-quality drugs, the functionalist ideal collapses.
➤ The pharmaceutical industry is failing in its social role — putting profits before people, in direct violation of the normative expectations of a welfare society. State, Regulation, and BiopoliticsThe revised Schedule M emphasizes quality control, data transparency (ALCOA+), and pharmacovigilance. But why were such reforms delayed until a crisis forced them? Michel Foucault – Biopolitics and Governmentality: Foucault explained how modern states exercise control over bodies and populations through subtle regulatory mechanisms — what he termed biopolitics.
➤ Biopower is only effective when state capacity is matched with political will. In India, fragmented regulation and bureaucratic inertia often render such controls reactive rather than preventive. Global Capitalism and the “Pharmacy of the World”India is often celebrated as the “Pharmacy of the Global South”, exporting generic drugs to over 200 countries. But this global reach raises ethical concerns. Immanuel Wallerstein – World Systems Theory: India occupies a semi-peripheral role in the global pharmaceutical system — producing cheap generics for both domestic use and export, often under pressure to cut costs and scale fast.
➤ Pharmaceutical safety reflects core-periphery dynamics — where profit flows north, and risks stay south. Corporate Power, Ethics, and Risk SocietyThe inclusion of Quality Risk Management (QRM) and pharmacovigilance in Schedule M reflects a growing concern with systemic risk in modern societies. Ulrich Beck – Risk Society: Beck argued that modernity creates new, manufactured risks (e.g., toxic chemicals, drug side effects) that are technologically complex and often invisible until disaster strikes.
➤In the risk society, science and technology do not just solve problems — they also create new ones, especially when corporate accountability is weak. Policy, Public Trust, and Structural ViolenceEvery regulatory failure reduces citizen trust in institutions. In India, where public health infrastructure is already under strain, such tragedies create public fear and resentment. Johan Galtung – Structural Violence: Galtung defined structural violence as harm caused by unjust systems — not through bullets, but through neglect, inequality, and inaction.
➤Drug safety is a moral issue, not just a technical one — and the failure to protect the vulnerable is a form of institutionalised violence. Key Features of Revised Schedule M – Sociological Interpretation
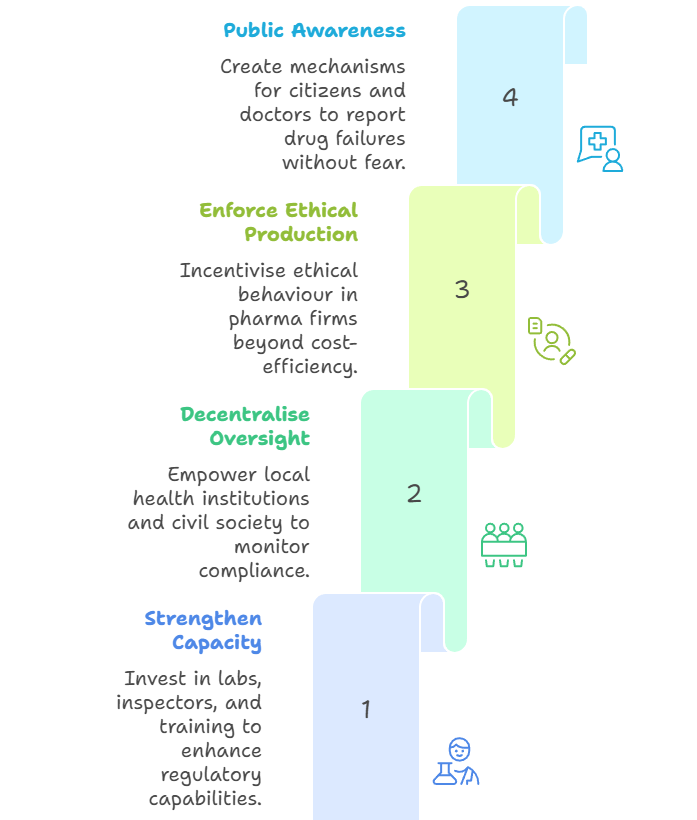
Way Forward: A Sociologically Informed Public Health Framework
Make Health a Right, Not a Commodity: Align pharmaceutical regulation with universal health ethics, not just market logic. |
To Read more topics, visit: www.triumphias.com/blogs
Read more Blogs:
Governing the Global Body: A Sociological Reading of the Amended International Health Regulations
The Startup Mandala: A Sociological Decoding of India’s New Caste System


8 comments