Children, Capital, and Control: A Sociological Lens on India’s Paediatric Drug Regulation Crisis
(Relevant for Sociology Paper 2: Population Dynamics)
|
In 2025, India once again found itself confronting a familiar tragedy: the death of 25 children in Madhya Pradesh due to contaminated cough syrup. These deaths, following earlier incidents in The Gambia and Uzbekistan linked to Indian-made syrups, reveal a persistent and structural crisis in paediatric drug regulation. While policy responses—ranging from tighter export norms to proposed surveillance portals—dominate official discourse, a sociological reading reveals something deeper: a crisis of governance, ethics, and social responsibility in how India regulates the health of its most vulnerable citizens. The Socio-Legal Architecture: Rights and Responsibilities
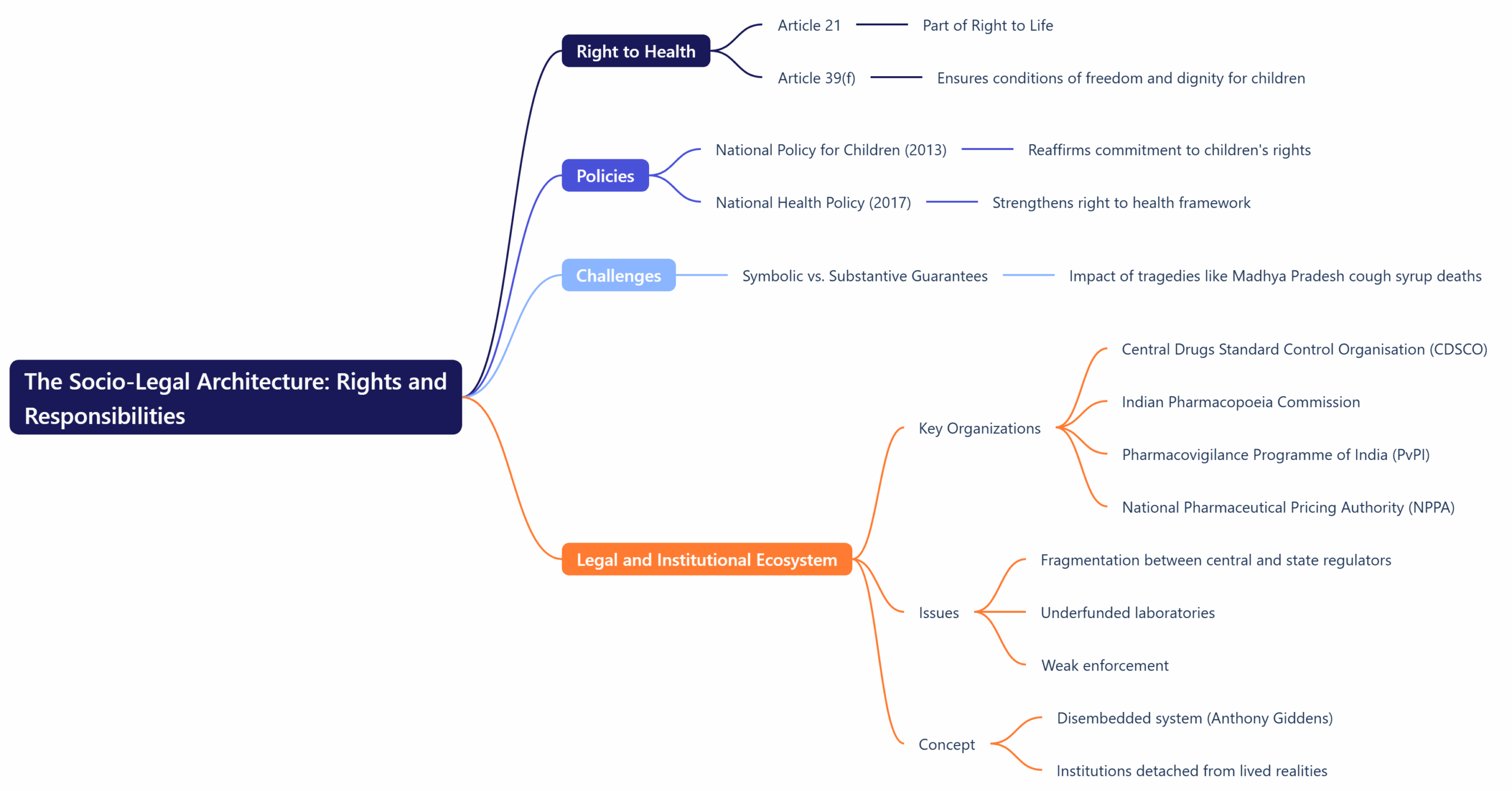
India’s constitutional framework enshrines the right to health as part of the Right to Life (Article 21), while Article 39(f) directs the State to ensure that children grow up in conditions of freedom and dignity. Policies like the National Policy for Children (2013) and National Health Policy (2017) reaffirm that commitment. Yet, when confronted with tragedies like the Madhya Pradesh cough syrup deaths, these guarantees appear symbolic rather than substantive. The legal and institutional ecosystem—the Central Drugs Standard Control Organisation (CDSCO), Indian Pharmacopoeia Commission, Pharmacovigilance Programme of India (PvPI), and National Pharmaceutical Pricing Authority (NPPA)—forms the backbone of drug governance. However, fragmentation between central and state regulators, underfunded laboratories, and weak enforcement have created what sociologist Anthony Giddens might call a “disembedded system”: institutions designed for modern governance but detached from the lived realities they are meant to serve. Power, Knowledge, and Surveillance: Foucault’s Medical GazeThe French thinker Michel Foucault provides a sharp lens for understanding how power operates in the realm of health. His concept of the “medical gaze” describes how modern institutions regulate bodies through observation, classification, and control. In India’s pharmaceutical governance, the medical gaze is paradoxically inverted. Surveillance is strongest at the level of export quality—driven by global scrutiny and economic pressure—while domestic oversight remains comparatively weak. This two-tier regime of drug safety, where international consumers receive safer products than Indian children, exposes a hierarchy of value: the bodies of Indian children are rendered less visible within the regulatory gaze. This echoes Foucault’s critique that modern biopower—while appearing humanitarian—often operates through differential visibility and control. The Political Economy of Child Health: Capital and RiskIndia’s pharmaceutical sector is a global powerhouse, exporting drugs to over 200 countries. Schemes like the Production Linked Incentive (PLI) for pharmaceuticals, Bulk Drug Parks, and Make in India have boosted industrial growth. Yet this economic expansion coexists with moral and regulatory negligence. From a sociological standpoint, this reflects Karl Marx’s notion of commodity fetishism: health itself becomes commodified, valued not for its social purpose but for its market utility. In a neoliberal framework where the pharmaceutical industry is both a source of national pride and foreign revenue, children’s health rights risk being subordinated to commercial imperatives. Ulrich Beck’s “risk society” concept further illuminates this paradox. Beck argued that modern societies, even as they advance technologically, generate new forms of risk—manufactured by institutions themselves. India’s repeated drug contamination crises are a textbook case: the pursuit of efficiency and profit generates systemic vulnerabilities, from adulterated ingredients to corrupt licensing. These are not “accidents,” but structural risks embedded within the very logic of industrial modernity. Institutional Fragmentation and the Sociology of Neglect
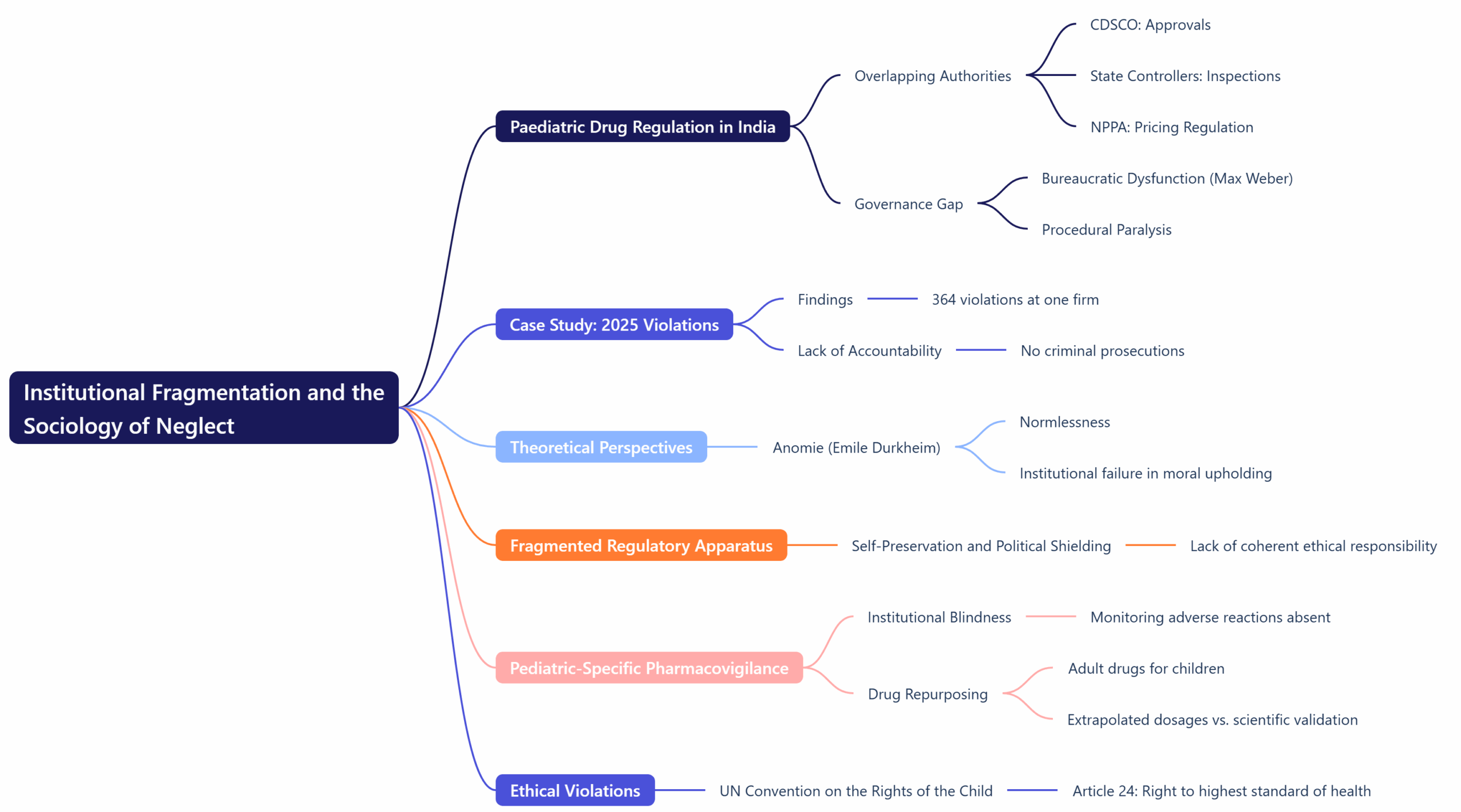
India’s paediatric drug regulation landscape is characterized by overlapping authorities and diffused accountability. The CDSCO oversees approvals, state controllers handle inspections, and agencies like the NPPA regulate pricing. The result is a “governance gap”—a phenomenon sociologist Max Weber might associate with bureaucratic dysfunction, where rational-legal authority devolves into procedural paralysis. The 2025 case revealed 364 violations at a single pharmaceutical firm, yet no official faced criminal prosecution. Here, Emile Durkheim’s concept of anomie—a state of normlessness where institutions fail to uphold collective morality—becomes relevant. The regulatory apparatus, rather than embodying shared ethical responsibility, often functions as a fragmented field of self-preservation and political shielding. Moreover, the lack of pediatric-specific pharmacovigilance—a system for monitoring adverse drug reactions in children—represents institutional blindness. Drugs tested on adults are routinely repurposed for children, with dosages extrapolated rather than scientifically validated. This practice violates both ethical norms and the UN Convention on the Rights of the Child (Article 24), which guarantees every child “the enjoyment of the highest attainable standard of health.” The Sociology of Inequality and Medical AccessIndia’s child health crisis is not only regulatory—it is social. The majority of pediatric medicine purchases occur over the counter, often without prescriptions. In slum communities and Tier-2 towns, parents administer doubled doses or mix formulations, unaware of brand variations in concentration. Pierre Bourdieu’s theory of cultural capital explains this dynamic. Access to health literacy—understanding dosage, reading labels, recognizing symptoms—is unequally distributed across social classes. The state’s failure to ensure caregiver education perpetuates what Bourdieu termed symbolic violence: the imposition of harm through systemic ignorance and inequality. This imbalance is amplified by market behavior. Branded paediatric drugs, marketed as superior, often remain inaccessible to the poor, while cheap generics—sometimes substandard—circulate widely. Thus, India’s pharmaceutical market reproduces the same class hierarchies that shape education, nutrition, and healthcare access. Ethics, Reform, and the Path ForwardThe proposed reforms—such as establishing a National Paediatric Drug Safety and Ethics Authority (NPSEA), creating a Paediatric Clinical Research and Data Ecosystem, and implementing QR-based batch tracking—reflect an emerging recognition that technological governance alone cannot ensure justice. Sociologically, the question is not only how to regulate, but for whom regulation serves. If governance remains confined to technocratic fixes—new committees, digital audits, and fiscal incentives—it risks reinforcing what Habermas called the “colonization of the lifeworld”: the intrusion of bureaucratic logic into domains of moral responsibility. True reform must integrate public participation, ethical transparency, and social accountability. Mandatory pharmacist training, caregiver education campaigns through Anganwadis and schools, and public disclosure of inspection reports would bring health governance back into the social domain—where citizens, not just corporations, become agents of oversight. Children as Citizens, Not SubjectsA transformative approach would treat children not as passive recipients of care but as citizens with rights. The National Policy for Children (2013) declared “Survival, Health, and Participation” as the three pillars of child rights. Yet, in practice, participation is missing from India’s pharmaceutical governance. A child-rights perspective would involve consultative mechanisms with pediatricians, caregivers, and advocacy groups in regulatory decisions—embodying what Giddens termed the “dialogic democracy” of late modernity. Regulation, then, becomes not a vertical act of control but a horizontal process of social negotiation. Conclusion: From Tragedy to TransformationThe deaths in Madhya Pradesh are not isolated lapses—they are symptoms of a structural disorder within India’s developmental modernity. Economic growth and pharmaceutical success have not translated into ethical governance or social justice. A sociological understanding reveals that reforming paediatric drug regulation is not merely a technical task; it is a moral and political imperative. As Durkheim wrote, society’s health depends on its collective conscience. When children die from preventable causes, it signals not just a market failure, but a moral failing of the state. India’s challenge is to move from reactive regulation to proactive care, from fragmented control to integrated accountability, and from seeing children as dependents to recognizing them as bearers of rights. Only then can India truly realize the World Health Organization’s vision: “Every child deserves safe, quality healthcare from the very beginning.” |
To Read more topics, visit: www.triumphias.com/blogs
Read more Blogs:
Faith, Privacy, and the Self: The Supreme Court’s Moral Turn in Defending Religious Autonomy
Global Knowledge, Local Futures: A Sociological Lens on India’s Opening to Foreign Universities


3 comments